Introduction
The periodic table of elements is far more than a classroom wall hanging; it is the master code of the physical world. Since Dmitri Mendeleev first published his table in 1869, it has evolved from a speculative arrangement into a precise predictive tool used by scientists to engineer everything from life-saving pharmaceuticals to the quantum processors of 2026. Every row (period) and column (group) tells a story of atomic structure, electronegativity, and reactive potential.
As we navigate the mid-2020s, our relationship with the periodic table is shifting. We are no longer just cataloging elements; we are entering an era of "Atomic Precision," where we manipulate specific elements to solve global crises. Whether it is the frantic search for Lithium to fuel the electric vehicle (EV) revolution or the use of Rare Earth Elements (REEs) in advanced defense systems, understanding these building blocks is essential for literacy in the 21st century.
Background / Context: The Architecture of the Table
Before diving into specific elements, it is vital to understand the "Operating System" of the periodic table. The table is organized by Atomic Number (the number of protons in the nucleus).
- Groups (Columns): Elements in the same column share similar chemical properties due to their valence electrons. For example, Group 1 (Alkali Metals) are all highly reactive, while Group 18 (Noble Gases) are famously stable.
- Periods (Rows): These represent the number of electron shells an atom has. As you move from left to right across a period, elements transition from metallic and highly conductive to non-metallic and insulating.
In 2026, the International Union of Pure and Applied Chemistry (IUPAC) remains the ultimate arbiter of the table. While the table currently ends at Oganesson (Element 118), research into the "8th Row" is a primary scientific trend, with laboratories in Japan, Russia, and the U.S. racing to discover Element 119 and 120.
The "Life-Givers": Essential Biological Elements
You cannot understand the periodic table without knowing the elements that compose over 96% of the human body. These are the "Biological Summits."
Carbon (C) – Atomic Number: 6
Carbon is the "Invention Element" of life. Its unique ability to form four stable bonds allows it to create the complex chains and rings found in DNA, proteins, and carbohydrates. In 2026, Carbon is also a central focus of climate science. We are moving from a "Carbon-Combustion" society to a "Carbon-Capture" society, where we use the element to create new materials like graphene.
Oxygen (O) – Atomic Number: 8
Oxygen is the engine of cellular respiration. While it is the third most abundant element in the universe, its presence in our atmosphere (21%) is what allows complex life to exist. In 2026, Oxygen research is critical for "Sustainable Aviation Fuels" and deep-space life support systems.
Nitrogen (N) – Atomic Number: 7
Nitrogen makes up 78% of our air and is a crucial component of amino acids. The "Haber-Bosch" process, which pulls Nitrogen from the air to create fertilizer, is what allows us to feed 8 billion people. However, in 2026, managing the "Nitrogen Cycle" is a major environmental challenge, as runoff from these fertilizers creates ocean "dead zones."
The "Tech-Drivers": Critical Minerals of 2026
In the current decade, the geopolitical and economic power of a nation is often determined by its access to specific, high-tech elements.
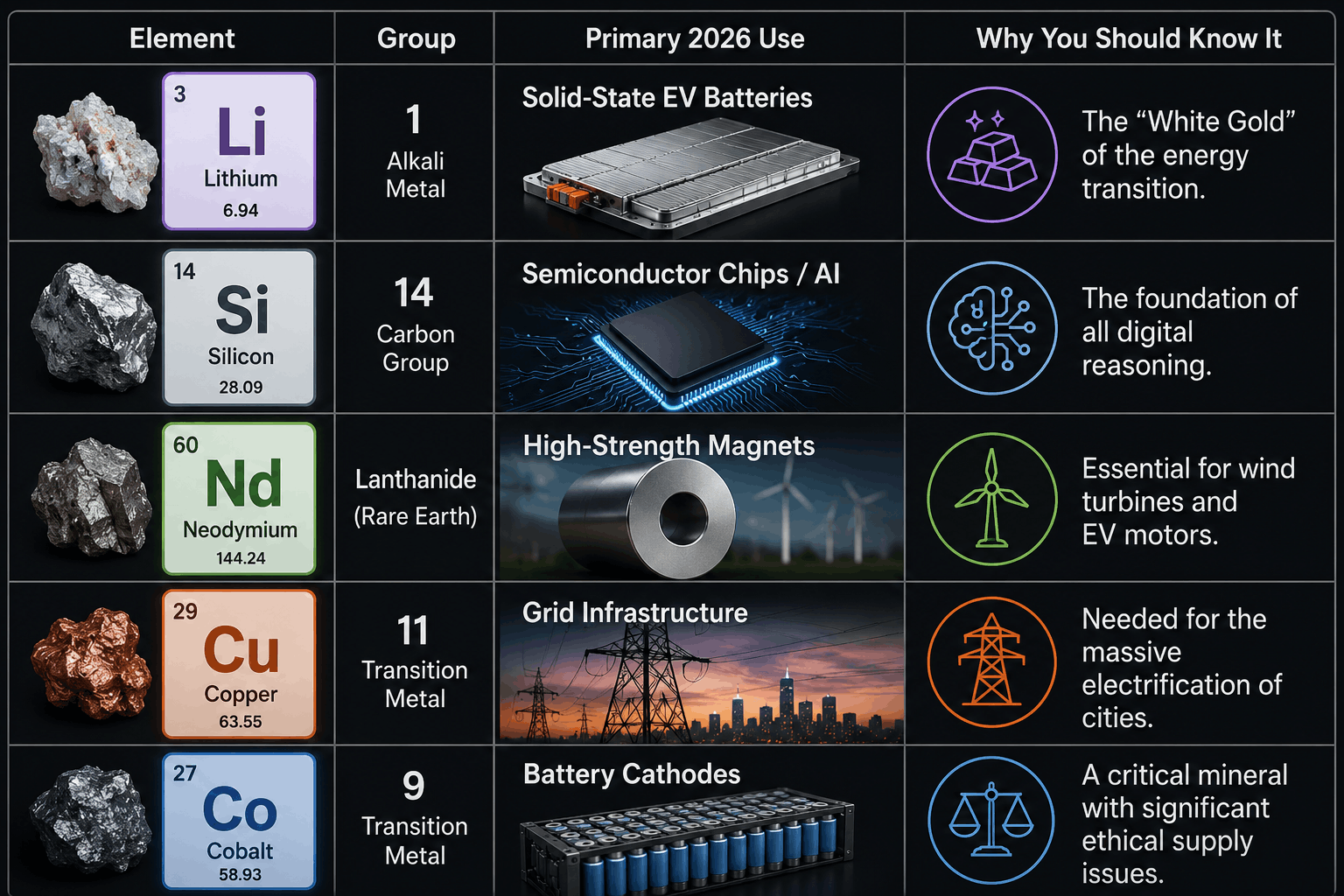 Tech-Drivers: Critical Minerals of 2026
Tech-Drivers: Critical Minerals of 2026
Lithium: The Energy Storage King
In 2026, Lithium-ion technology has hit its "Peak Efficiency." We are now seeing the rise of Solid-State Lithium batteries, which are 50% smaller and far safer. Lithium demand is projected to grow by 16% annually through the late 2020s, making it the most strategically important alkali metal on the table.
Neodymium: The Magnetism Master
Part of the "Rare Earth" family, Neodymium is used to create the world's strongest permanent magnets. In 2026, these magnets are the "Muscles" of modern tech, found in everything from the vibrating motors in your smartphone to the massive generators inside offshore wind turbines.
Analysis / Discussion: The "Rare Earth" Geopolitics
In 2026, the term "Rare Earth Elements" (REEs) is a frequent headline. These 17 elements (the Lanthanide series plus Scandium and Yttrium) are not actually "rare" in the Earth's crust, but they are rarely found in high concentrations and are difficult to refine.
- The Monopoly Problem: As of 2026, the supply chain for refined REEs is highly concentrated, with a single region controlling over 90% of global magnet production.
- The Defense Aspect: Modern guided weapons systems use up to 18 different critical minerals. A Tomahawk missile or an F-35 fighter jet cannot function without these specific chemical signatures.
- The 2026 Debate: Critics argue that our reliance on these elements is a "Progress Trap," leading to destructive mining practices. Proponents point out that without them, a "Green Transition" is physically impossible.
 The Rare Earth Elements and Their Spectra
The Rare Earth Elements and Their Spectra
Benefits / Importance: Elements as Climate Sentinels
The periodic table is our best tool for environmental restoration.
- Catalysts for Hydrogen: Elements like Platinum (Pt) and Iridium (Ir) are essential for electrolyzers that create Green Hydrogen—a clean fuel that emits only water vapor when burned.
- Carbon Capture Materials: New 2026 breakthroughs in Metal-Organic Frameworks (MOFs) use ions of Zinc or Copper to "sponge" CO2 directly out of the atmosphere.
- Molecular Editing: Chemists in 2026 are using "Molecular Editing" to swap individual atoms in a molecule’s core, creating biodegradable plastics that break down into harmless monomers like PET.
Challenges / Issues: The Ethics of Extraction
While these elements provide incredible benefits, their extraction is the primary ethical crisis of 2026 chemistry.
- "Urban Mining": Because traditional mining of Cobalt and Nickel is so environmentally taxing, 2026 has seen a massive boom in "Urban Mining"—recycling elements from old smartphones and EV batteries.
- Tailings Toxicity: The chemical separation of Rare Earths produces hazardous waste. In 2026, new "Hydrometallurgical" technologies are being regulated to ensure that mining does not contaminate local water tables.
- The Isotope Disruption: Modern medicine uses specific isotopes (like Technetium-99m) for cancer imaging. In 2026, supply chain disruptions for these isotopes are causing global delays in diagnostic procedures.
Future Outlook / Trends: AI and the "Post-Oganesson" Era
What does the horizon look like for the periodic table beyond 2026?
- AI-Discovered Elements: AI models are now predicting "Stable Isotopes" that haven't been synthesized yet, suggesting an "Island of Stability" where superheavy elements might last for years instead of milliseconds.
- The 8th Row: Scientists are currently attempting to synthesize Element 119 (Ununennium). If successful, this would be the first element in a new row of the table, potentially possessing properties that defy our current understanding of Group 1 chemistry.
- Synthetic Biology: By 2030, we may see the invention of "Xeno-Biology," where organisms are engineered to use non-standard elements (like Arsenic or Selenium) in their core metabolic processes.
Conclusion
The periodic table is the ultimate map of our planet's physical limits and its infinite potential. Whether it is the ancient, foundational role of Carbon or the 2026 strategic importance of Neodymium, each element is a key that unlocks a different part of our future. As we continue to refine our mastery over these building blocks, we must balance our desire for technological conquest with a deep respect for the Earth's finite resources. In 2026, the table is no longer a static chart it is a living, breathing guide to the survival of our species.
References & Data Sources
- American Chemical Society (ACS) - 2026 Element Data and Periodic Trends.
- IUPAC Isotopic Abundances Commission - Official Atomic Weight Revisions (2025).
- International Energy Agency (IEA) - The Role of Critical Minerals in Clean Energy Transitions (2026 Report).
- PubChem - NIH - Machine-Readable Element Property Database.
Want more practice?
Take today's free daily challenge — a new set of questions every 24 hours.
Start Today's Challenge Browse All Quizzes